Pipeline
Clinical Trials
Drug A
Drug B
Drug C
PRECLINICAL
IND ENABLING
PHASE I
PHASE 2A
PIVOTAL
Robust Data Package Supports Transition To The Clinic
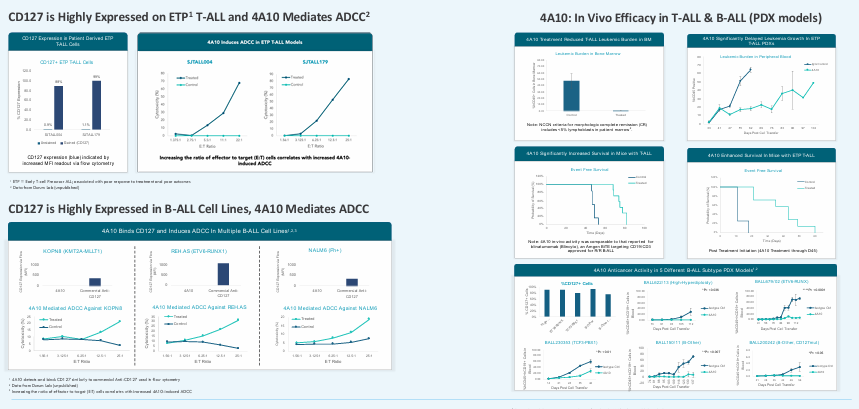
Development History : Progress To-Date
Substantial Non-Dilutive Capital Secured To De-Risk Early Development
- 1Q25: Completed Preclinical Proof-of-Concept, IND Enabling Studies, and Key CMC Work
- 3Q25: FDA Cleared 4A10 Investigational New Drug Application (IND)
- 4Q25-1Q26: Transition to the Clinic and Initiate the Phase 1 Clinical Trial

